January 10, 2021
Pakistani drug companies rarely bring innovative products to market. Ferozsons broke the mould
The company’s partnership with Gilead Sciences has resulted in better products available to the Pakistani market, and a substantially better financial performance for the company itself

Taimoor Hassan
5 views

The Pakistani pharmaceutical has its fair share of issues: a lack of innovative products that the industry blames on regulatory bottlenecks and price controls and depressed profitability as a result. While there are quite a few domestic companies that are doing well, in the industry, there are few stars and fewer still that have performed well financially by bringing access to innovative products for Pakistanis.
Ferozsons Laboratories is the exception. The company signed an agreement in 2014 with American life sciences company Gilead Sciences to introduce a breakthrough drug for Hepatitis C in Pakistan. Sovaldi, the drug to cure Hepatitis C, was introduced in Pakistan in 2014 by Ferozsons. Previous treatments for Hepatitis C only treated its symptoms and assumed it was a chronic disease a person had to live with. Sovaldi changed that, making it a curable disease.
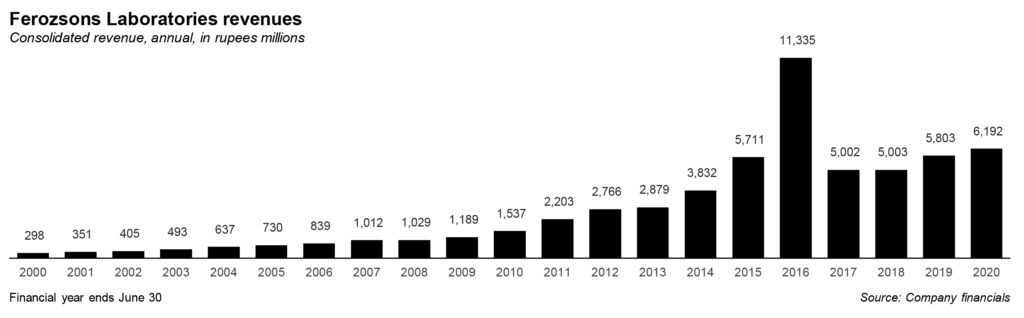
Following the launch of Sovaldi in Pakistan just a few months after it first launched in the United States itself, Ferozsons made a killing: revenue swelled to a high of Rs11.3 billion in 2016, from Rs3.8 billion just two years earlier. While the increase in revenue from that drug did not last, the company is hoping that it can continue to rely on Gilead’s innovation pipeline to provide new products to Pakistanis who need them.
In 2020, for instance, the disease ravaging the whole world is Covid-19 and one of the most effective drugs against it is Remdesivir. Like in the case of Sovaldi, the partnership again is with Gilead Sciences for production of Remdesivir in Pakistan by Ferozsons. And once again, the drug might be a lifeline for the company to improve revenues and profits.
The pharmaceutical industry in Pakistan is highly regulated, with the government fixing drug prices. Consequently, margins have remained relatively thin for the entire pharma industry, with many companies exiting the market completely due to high costs of production and insufficient profitability. In such a market, partnership with Gilead has come handy for Ferozsons. [restrict paid="true"]
The Gilead partnership
Not too long ago, Hepatitis C, considered an epidemic in the public health landscape of Pakistan, used to be considered insurable, and a chronic disease that was very difficult to treat. According to the World Health Organisation (WHO), viral hepatitis is the eighth highest cause of mortality globally and was responsible for an estimated 1.34 million deaths in 2015, a toll comparable to that of HIV and tuberculosis.
Globally, approximately 257 million persons are chronically infected with Hepatitis B and 71 million with Hepatitis C. At this rate, the WHO has estimated that between 2015 and 2030, an estimated 20 million deaths will occur because of Hepatitis.

Within the broader Middle East, North Africa and South Asia (MENASA) region, Pakistan and Egypt bear 80% of the disease burden and within Pakistan, almost 12 million people are suffering from Hepatitis B or C. Each year, about 150,000 new cases of patients with Hepatitis emerge, making Pakistan one of the largest populations in the world with this disease.
For years, the antiviral drug Interferon has been used as standard treatment for Hepatitis C, prescribed usually together with another antiviral medicine Ribavirin. Because it affected such a large population in Pakistan, Ferozsons had been producing drugs for treatment of Hepatitis B and C since 2003, initially importing the aforementioned Interferon injections.
From imports, the company moved on to set up a plant in Lahore as a joint venture with an Argentine partner to produce Interferon injections locally because the drug was expensive to import and difficult to supply in the market because it required cold chain conditions, that is, a temperature-controlled supply chain that requires an uninterrupted series of refrigerated production, storage and distribution activities, along with associated equipment and logistics to maintain quality achieved through keeping the drug at a desired low-temperature range.
“We were the first company to start producing interferons locally, in 2009,” says Osman Khalid Waheed, the Harvard-educated CEO of Ferozsons Laboratories. Waheed completed an undergraduate degree in economics from Harvard in 1993.
Interferons were not an easy drug. The entire course of treatment spanned six months to a year, with roughly 60% cure rate and numerous side effects, making it not the ideal drug but the only one that was reasonably effective in treating the disease. By year end 2013, California-headquartered Gilead Sciences launched Sovaldi – a breakthrough drug in Hepatitis C disease management with promising results. Sovaldi cured patients in a much shorter duration than interferons, with 90-96% cure rate and fewer side effects.
“Sovaldi was the product of Gilead Sciences. It was the first treatment approved by the [United States] Food and Drug Administration (FDA) for the treatment and cure of Hepatitis,” says Osman.
When this technology shift began happening, it was a welcome occurence of course because it was a big pain point for Hepatitis patients globally and in Pakistan to go through agonising treatment of six months to a year of Interferon injections.
For Ferozsons, however, the challenge was existential because the company had made a major investment to set up the aforesaid plant to manufacture interferons, dedicated entirely to making these injections that were about to become obsolete after the discovery of Sovaldi. This naturally prompted Ferozsons to seek partnership with the company [Gilead] that was going to disrupt liver disease management, and initiated efforts to that effect.
“Ferozsons very eagerly started pursuing a relationship with Gilead. Through some specialists in liver disease, who knew our company and the work we were doing hepatology (the branch of medicine concerned with the study, prevention, diagnosis and management of liver disease) in this part of the world, and who also knew Gilead, we got an initial introduction. There was an affinity between the two companies from the beginning because both had a very similar approach to disease management, to patient centricity, to making sure that access is central to what we do,” the affable CEO of Ferozsons told Profit.
The partnership was consummated in 2014 with the launch of Sovaldi in Pakistan. Osman underscores that the partnership was remarkable in many perspectives because normally, the history of big pharma suggests that when a company discovers a drug, the focus primarily is to offer the new drug in the high-income countries such as in the West.
“These are the markets that have healthcare systems that can afford expensive treatments. The treatments are expensive. Globally, the cost of developing any drug can go over a billion dollars because of massive research and development expenses, expenditure on processes, final approval and making the drug available in the market,” explains Osman.
Only when the drug supply is sufficient for these countries does Big Pharma turn its focus towards developing countries, which normally have a gap of a few years; five to ten years according to Osman. However, in Sovaldi’s case, the drug was made available in Pakistan in a few months because of the partnership that was nearly unprecedented.
“In the case of Sovaldi, within a month of FDAs approval, Gilead began a series of access partnerships across the developing world. Less than a year after the approval of the drug in the US, the first patient in Pakistan was put on treatment,” says Osman.
What was definitely unprecedented, and surprising, was that besides expediting the availability of physical access to the drug, the drug was made available in Pakistan at a ridiculously low price.
In the US, Sovaldi became controversial because it had been priced using the principle that how much a patient suffering from chronic Hepatitis C disease would save if he was put on the novel drug. The savings were substantial and that reflected in the price of a single pill of Sovaldi, priced ludicrously at $1,000.
While the healthcare system in the US, where health insurance covers medicine costs, nobody was going to be able to afford that kind of pricing in Pakistan. For countries like Pakistan, Gilead created a special access programme where companies like Ferozsons were able to offer the drug at 2% of its price in the United States. The drug was priced high in countries that could afford such prices and from these markets, Gilead would recover expenses incurred to make the drug.

“Initially, it was a Gilead product. Later, we received a license to produce our own generic. That brought down the treatment of Hepatitis C down to under Rs10,000 for a patient on Sovaldi for a three month course, against the Interferon course that, when it was imported, used to be a treatment of Rs300,000 for a patient for six months to a year and that is a remarkable improvement for a patient,” says Osman.
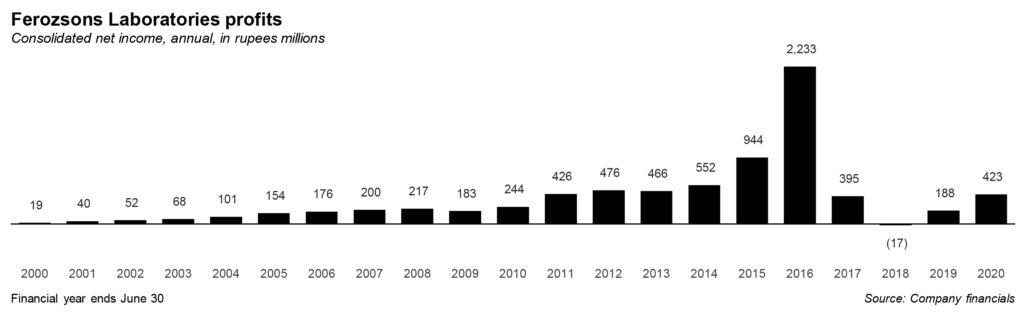
Hepatitis C is pervasive in Pakistan and the statistics in this regard are depressing. Therefore, though margins were thin because of low prices, the company was able to make large profit after tax, over Rs2 billion, on the back of large sales volume. According to an estimate provided by the CEO, Ferozsons helped cure 70,000 patients suffering from Hepatitis C in the first year alone of the launch of the drug in Pakistan.
Things, however, are not easy in Pakistan. In 2017, sales demonstrated a decline of 58% owing majorly due to a decrease in, mainly Gilead-licensed Sovaldi. A year after sales of Sovaldi began, the federal government granted unlicensed generics entry into the market. Where Sovaldi was a brand name licensed with Gilead, the original medicine was Sofosbuvir. So drugs with similar chemical composition as the original drug were granted entry into the market. And hence the fall in profits. From Rs2 billion, company’s profits in the next year were Rs395 million only.
The CEO candidly said that when generic drugs to treat Hepatitis C were granted entry into the market, Sovaldi was almost bigger than the rest of the company. “We have normally had a positive bottom line but if you compare that to that one period, because the volume of that business was large, the trends went down,” says Osman.
Like in the case of interferons, large investments were made by the company to produce Sovaldi locally but with generics making entry into the market, it was soon a lost investment.
From Sovaldi to Remdesivir
When Covid-19 hit, the go-to drug, in the absence of a clinically proven cure that had the chances of inhibiting the Covid-19 infection was a drug sold under brand name Veklury, a broad spectrum antiviral medicine developed by Gilead Sciences originally to cure Ebola virus. The drug is more commonly known by its generic name Remdesivir.
Luck played its part that Remdesivir turned out to be the only drug that was effective against Covid-19 and luck played its part that it was Gilead that had developed the drug. Ferozsons was lucky that it had been a partner with Gilead for a few years and the partnership paid off when Gilead granted license to Ferozsons among five companies, four of which were from India, to expand the supply of Remdesivir in the region.
The partnership was rather easy to win because of earlier collaboration between Gilead and Ferozsons to produce Sovaldi in Pakistan. Though other producers of Remdesivir were also available, Ferozsons was the only company licensed by Gilead, the original producer of Remdesivir, to sell the drug in Pakistan. Ferozsons sells Remdesivir under the brand name Remidia in Pakistan.
“Generally, Remdesivir is a difficult product to manufacture so that was a challenge. More importantly, because of the urgency of the pandemic, the technology transfer had to take place in a matter of weeks. So things were done really quickly. It is an important development in the pharma industry because this is the first time that a producer in Pakistan was made part of an international supply partnership,” Osman told Profit.
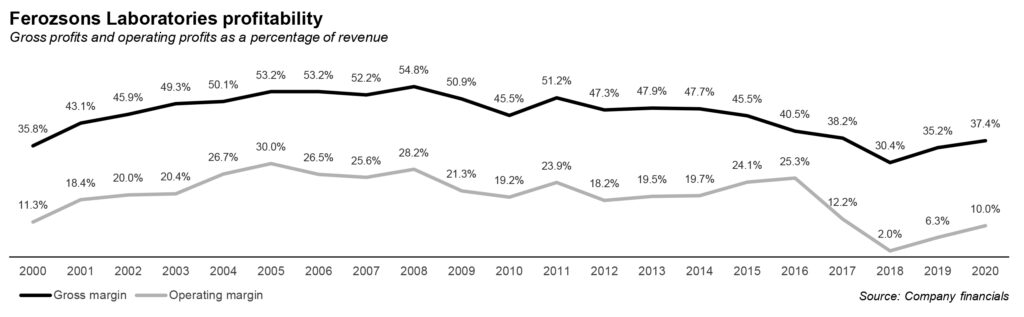
The impact on revenue and profits has been visible. For the year 2020, Ferozsons’ revenue has crossed Rs6 billion mark from its steady range of Rs5 billion to under Rs6 billion between 2015 and 2020, except in 2016. The revenue has remained stable between these years and partnership with Gilead can be considered a spike in the business for the company. However, the bottomline shrunk due to the impact of the drop in Sovaldi sales as noted above.
However, in the case of Remdesivir, the spike might not be profound. In the case of Remdesivir as well, margins are thin but volumes are better because of the export opportunities. When Gilead licensed Ferozsons for local supply of Remdesivir among five other companies, the license allowed Ferozsons and others to distribute their respective brands of the drug to other low-income countries as well and that has opened up foreign markets for Ferozsons.
“We have been supplying Remdesivir to Ukraine and Indonesia and both the countries are highly regulated markets that normally do not buy medicines from Pakistan. We are looking at this as a window of opportunity to establish ourselves in these markets through the quality approvals that we were getting in the process,” says Osman.
“It has opened up markets for us. We are looking at Remdesivir as a bridge rather than as a money making opportunity,” the CEO said.
The use of Remdesivir is going to decline as vaccines become widely available. In the hyperspeed context of things, the vaccines to cure Covid-19 are going to be available very soon and the focus of companies like Ferozsons is going to shift towards producing these vaccines for domestic supply, might as well for exports in the markets that opened up with the Remdesivir license from Gilead.
A stifling regulatory environment
Innovation in the pharmaceutical industry in Pakistan has been figmental. There is a reason why all the breakthrough drugs are developed in the Western countries, or China for example. Research and development in the pharmaceutical industry in Pakistan is near to none owing to regulatory frameworks that stifle innovation and lack of critical mass of scientists.
Unfortunately, the scientific base in Pakistan is too small to start real innovation in basic chemistry and basic sciences on a large scale; the scale of producing a drug to cure a disease in a country of 200 million people. The research and development required for innovation in medicine is further dragged down by a difficult regulatory status-quo that finds it convenient to deny permission to any new innovation because of its own capacity issues. Henceforth, for the pharmaceutical industry, it has been a discouraging environment for any innovation.
Though a partner of Gilead for many years, there has been no collaboration between Ferozsons and the US company on the research front because big research companies can not really have Pakistan on their radar when the regulatory environment is not conducive to research. And those that were researched and developed by the company on their own, have hit a rock bottom after a Supreme Court judgement that mandated Cabinet approval for all drugs.
“It has been a very discouraging environment for any clinical trials etc to take off. Next door in India, clinical trials are big export earners for the country. In Pakistan, they are almost non-existent. So when you can not have clinical trials, it means big research companies can not really have Pakistan on their radar. Before Remdesivir, foreign companies did not have Pakistan on their manufacturing radar either,” says Osman.
The problem at the end of the day, not only for Ferozsons but the entire pharmaceutical industry in Pakistan, is that capacity issues have always kept focus of regulators on controlling prices of drugs instead of creating progressive regulatory framework for innovation in the pharma industry. Pharma industry in Pakistan is regulated under The Drugs Act, 1976 and Drug Regulatory Authority of Pakistan Act (DRAP), 2012. The acts governing the pharma industry vest the authority to control prices in the federal government of Pakistan.
Drugs generally are a very politicised subject and in Pakistan, there is not enough emphasis placed on the fact that it is the government’s responsibility to cater to the health and education for the large population that they govern. The conventional thinking with the government is that cheap healthcare is best healthcare. That is why drug prices have been ridiculously low in Pakistan as compared to Western countries. Nobody thinks that there should be health insurance systems and if someone is ill, the state should step in and treat them like it happens in the rest of the world. In Pakistan, the government finds it more convenient that the responsibility should be left on the public and for political expediency, all the emphasis has been on the prices instead of drug quality, innovation and research and development.
For companies like Ferozsons, artificially low prices do not leave enough financial capacity in the company to venture into expensive research and development that leads to innovation and breakthrough drugs. And the de-facto authority to regulate prices in Pakistan has been DRAP that has thus far remained unempowered.
“DRAP needs to be autonomous, manned by a professional permanent CEO. Presently, DRAP officers are appointed on an ad-hoc basis for a three-month tenure. The government has not been able to work out long-term contracts for these officers. DRAP CEO is an acting CEO, DRAP directors are acting directors. Their appointment is done for 90 days which then lapses and they are appointed for another 90 days. So there is also a lag where the government sits on their reappointments and DRAP becomes completely toothless,” Osman says.
“Secondly, DRAP should focus on making sure that there is standardisation in the country because that is what their technical capabilities are. Pricing is the matter of mathematics and anyone can oversee that. There are technical issues that need to be addressed and that is what DRAP should be looking at. Overseeing prices only is a way of taking your attention away from what your actual job is. When you also know that every question is going to be about prices, then you forget about other technical responsibilities. Counterfeit drugs have been in the market. It has been reported but how much regulatory action have you seen pertaining to that? So all of that is possible if DRAP is given that role and then enabled to perform,” he adds.
Then there is Cabinet that now sits on drug approvals and that has become another pain for the industry. For about two years now, the Cabinet has not approved a single drug, as the CEO told us, that includes a drug by Ferozsons; a vaccine to cure Hepatitis E. Hepatitis E is an acute form of liver disease that can be deadly if people have other liver diseases. For pregnant women, it carries great risks with 25-30% mortality rate. Pakistan is also hyper endemic in Hepatitis E which means that the disease has the burden here more than other countries in the world.
But while things have been murky in the past, Osman hinted at a change in the mindset in the government after Dr Faisal Sultan, the incumbent advisor to the Prime Minister on health, assumed charge. Approvals have gone through the Cabinet after a dreadful delay and Ferozsons Hepatitis E vaccine has also been approved. With the approval, the company is hopeful of another surge in revenue and profits. But this time, it is going to be because of the drug developed by Ferozsons. [/restrict]

Taimoor Hassan
The author is a staff member and can be reached at taimoor.hassan@pakistantoday.com.pk







